
Published by Guangdong Keyao Purification M&E Engineering Co., Ltd. — 13 years of expertise in plant extract cleanroom engineering, serving 100+ clients.
Are You a Phytic Acid Sodium Manufacturer? How Many of These Problems Sound Familiar?
If you're in the phytic acid sodium business, have you encountered any of these situations:
Your EU-bound shipment gets stopped at the port — total aerobic count exceeds limits, the entire batch is destroyed, losses reach hundreds of thousands of yuan, and the client immediately suspends cooperation;
Packaged products arrive at the customer's facility already clumped together — the client calls demanding answers, and you have no idea where the problem originated;
Heavy metal tests pass every time, but lead content keeps hovering near the limit — you have no confidence, and when clients ask during factory audits, you can't provide a clear explanation;
Same formula, same production line — one batch tests at 99% purity, the next at 95% — clients start questioning your quality control;
EU clients request production environment validation reports and cleaning verification documents — you search the entire factory and can't produce a single proper document.
If two or more of these apply to you, this article is worth reading carefully.
These problems appear to be process issues on the surface, but the root cause is the production environment. The extraction process for phytic acid sodium isn't inherently complex, but once the production environment loses control, microorganisms, moisture, heavy metals, and foreign matter infiltrate from every direction, gradually eroding your purity, stability, and compliance credentials.
The "Invisible Killers" in Plant Extract Cleanrooms: Why Purity and Contamination Issues Keep Recurring
Many phytic acid sodium manufacturers share a common misconception: they attribute quality problems to raw materials or processes while overlooking that the production environment itself is the biggest source of contamination.
Insufficient Cleanroom Standards — Where Do Microorganisms Come From?
The raw materials for phytic acid sodium — rice bran, corn, and soybean meal — are natural breeding grounds for microorganisms. The total bacterial count in incoming materials can exceed 10⁶ CFU/g. When extraction, filtration, and drying processes take place in ordinary workshops, airborne bacteria continuously settle into materials. Products dried and then re-exposed before packaging face nearly unavoidable secondary contamination.
The more hidden problem: after drying, the product seems "safe," but the packaging workshop's air cleanliness is often worse than the extraction area — frequent personnel traffic, contaminated packaging materials, and HVAC systems that have never been cleaned. For many companies, microbial exceedances happen in this "last mile."
Moisture Absorption and Clumping — Rooted in Packaging Workshop Humidity Failure
Phytic acid sodium is highly hygroscopic. Exposed to relative humidity above 60%, it begins absorbing moisture within hours. Many companies' packaging workshops lack independent dehumidification systems. During the rainy season, humidity easily exceeds 80%, and products start absorbing moisture right on the packaging line. Once sealed, clumping continues inside the package. By the time customers receive the goods, the damage is irreversible.
Heavy Metals "Near the Limit" — A Dual Contribution from Equipment and Air
Phytic acid sodium has an extremely strong chelating ability for heavy metals — this is its functional characteristic, but it also means it "absorbs" metal ions encountered during production. Ordinary carbon steel or low-grade stainless steel equipment continuously leaches metal ions under corrosion from acidic extraction solutions. Metal dust in workshop air also settles into materials. Testing "just barely passing" every time isn't good process control — it's just that you haven't hit a raw material fluctuation yet.
Batch Purity Fluctuations — A Direct Reflection of Unstable Environmental Control
Same line, same formula, but purity swings between 95% and 99% between batches. The root cause is often not process parameters but rather: incomplete cleaning from the previous batch allowing residue contamination; workshop temperature and humidity fluctuations affecting crystallization; and cross-contamination from dust between batches. Environmental instability is the greatest enemy of batch consistency.
Five-Step GMP Cleanroom Construction Method for Phytic Acid Sodium: Solving All Four Problems at the Root
This is a systematic solution we developed after serving multiple phytic acid sodium and plant extract manufacturers. Rather than treating symptoms, it addresses the fundamental environmental engineering logic to build a plant extract cleanroom where "microorganisms cannot survive, moisture cannot invade, and heavy metals and foreign matter have nowhere to hide."
Step 1: GMP-Based Cleanroom Zoning Design for Phytic Acid Sodium Workshops
Risk-based contamination zoning with unidirectional personnel and material flow.
The workshop is divided into three zones:
- Core Clean Zone (Class 100,000): Crystallization, drying, grinding, packaging — processes where products are directly exposed, maintaining positive pressure ≥15Pa
- General Clean Zone: Extraction, ion exchange, concentration — materials contained in sealed equipment
- Non-Clean Zone: Raw material preprocessing, warehousing — physically separated from clean zones
Personnel purification follows a six-step procedure: shoe change → remove outer clothing → hand washing → don cleanroom garments → hand disinfection → air shower. Materials transfer through pass-through windows or sealed pipelines. Dust-generating areas (grinding, packaging) maintain relative negative pressure (-5 to -10Pa) to prevent dust from spreading to clean zones.
Step 2: Cleanroom Standards and Low-Humidity Environment Control
Keep moisture out and lock dryness inside the workshop.
Deep dehumidification units are integrated with clean air conditioning to control the packaging workshop dew point below -10°C (corresponding to approximately 30% relative humidity or less). Local dry gas protection hoods are installed at drying, grinding, and packaging stations, supplying HEPA-filtered dry hot air or nitrogen to create a micro-positive pressure protective barrier. Online dew point meters provide real-time monitoring with automatic alarms when limits are exceeded. All compressed air contacting products undergoes three-stage filtration for oil, water, and bacteria removal, with dew point ≤-40°C.
Step 3: Full-Process Heavy Metal Interception in GMP Plant Extract Facilities
Simultaneous action across three dimensions: equipment materials, air purification, and online detection.
- Equipment Selection: All equipment, piping, valves, and tanks contacting materials use 316L stainless steel with mirror-polished interior surfaces (Ra≤0.4μm), eliminating metal ion leaching at the source
- Air Purification: HVAC systems include chemical filtration stages (activated carbon + potassium permanganate-impregnated alumina) to adsorb heavy metal particles and VOCs from workshop air
- Online Detection: Metal detectors (sensitivity ≥Fe 0.5mm, SUS 1.0mm) and magnetic separators (≥10,000 Gauss) are installed after grinding and before packaging for 100% foreign matter rejection; ultrasonic vibrating screens ensure uniform product fineness
Step 4: Microbial Triple Defense System for Phytic Acid Sodium Production
Leaving microorganisms no gap to survive.
First Line of Defense — Environmental Air: Class 100,000 cleanroom with HEPA filters (filtration efficiency ≥99.97%), scheduled ozone/UV disinfection, regular airborne and settling bacteria testing, and established environmental monitoring archives.
Second Line of Defense — Equipment Cleaning: Sealed pipeline production to avoid manual material exposure. Online CIP cleaning systems automatically clean and disinfect extraction, concentration, and crystallization equipment. Cleaning verification procedures (swab sampling for microbial residue testing) ensure every cleaning is recorded, documented, and traceable.
Third Line of Defense — Drying and Sterilization: High-temperature vacuum drying (80-100°C) serves dual drying and sterilization functions. Pre-packaging material screening through vibrating screens (100 mesh or finer) provides the final physical barrier.
Step 5: Online Monitoring and Export-Compliant Full-Batch Traceability System
Every batch's production environment must leave a verifiable "environmental history."
Particle counters, airborne bacteria samplers, temperature/humidity sensors, differential pressure sensors, and dew point meters are deployed in core clean zones, with data fed in real-time to SCADA/MES systems and automatically linked to production batch numbers. Each batch automatically generates an "environmental history" containing cleanliness data, temperature/humidity curves, pressure differential records, and cleaning verification results — this document is the core confidence for EU client audits, FDA inspections, and BRC certification.
Real Case Study: How a Northern China Phytic Acid Sodium Factory Reclaimed EU Orders with a Single Cleanroom
A specialized phytic acid sodium manufacturer in Northern China, exporting to the EU and US, reached out to us when their Quality Director Mr. Wu had just experienced a devastating shipment return.
Before Renovation: Four "Disasters" in One Year
Microbial Contamination Return: An EU-bound batch of phytic acid sodium was found to exceed total aerobic count limits at port inspection. The entire batch was destroyed — a direct loss of 500,000 yuan. The EU client immediately suspended their supplier qualification for one year. The root cause was insufficient air cleanliness in the extraction workshop, compounded by secondary exposure in an ordinary packaging workshop after drying.
Product Clumping Complaints: During the rainy season, packaging workshop humidity reached 85%. Products clumped extensively within one month of packaging, with a customer complaint rate as high as 20%. Each complaint required re-grinding and re-packaging — double losses in cost and reputation.
Heavy Metal Warning: One batch's lead content tested close to the 10ppm limit. Although it didn't exceed the standard, the US client demanded a heavy metal control risk assessment report — something the existing workshop conditions simply couldn't support.
Failed Client Audit: An international pharmaceutical company conducted a factory audit. The auditor noted: no cleanroom classification, no temperature/humidity monitoring records, no cleaning verification procedures, no environmental monitoring data — directly rated as "unqualified supplier."
Mr. Wu said that during that period, what he feared most wasn't the test results — it was receiving a call from a client.
After Renovation: 18 Months of Data Speak for Themselves
Our company designed and built a Class 100,000 clean, low-humidity, full-process contamination-proof production workshop specifically for phytic acid sodium:
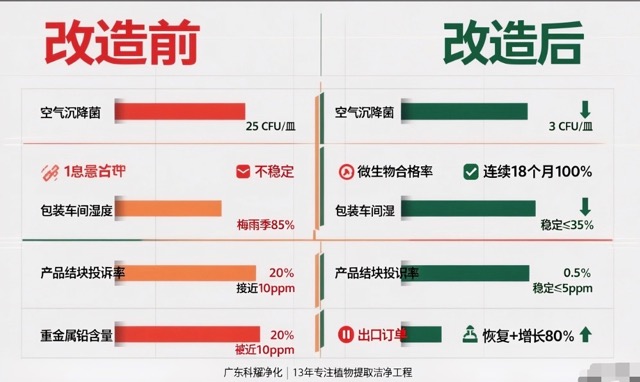
| Metric | Before | After |
|---|---|---|
| Air Settling Bacteria | 25 CFU/plate | 3 CFU/plate |
| Product Microbial Test Pass Rate | Unstable | 100% for 18 consecutive months |
| Packaging Workshop RH | Up to 85% in rainy season | Stable at ≤35% |
| Product Clumping Complaint Rate | 20% | 0.5% |
| Heavy Metal Lead Content | Near 10ppm | Stable at ≤5ppm |
| Foreign Matter Complaints | Frequent | Zero |
| Export Orders | Suspended | Restored with 80% growth |
In the most recent BRC certification audit, the cleanroom, environmental monitoring system, and cleaning verification procedures all received high scores. Mr. Wu's company successfully entered the qualified supplier lists of two international pharmaceutical giants.
"Before, we were gambling — betting that each batch would pass inspection. Now we're managing — managing the production environment for every batch."
— Mr. Wu, Quality Director
Guangdong Keyao — 13 Years of Expertise in Plant Extract Cleanrooms
Guangdong Keyao Purification M&E Engineering Co., Ltd., established in 2012 and headquartered in Nanhai District, Foshan, Guangdong Province. For 13 years, we have focused on providing complete cleanroom solutions for biopharmaceutical, plant extract, health supplement, food, and cosmetics industries, serving over 100 clients with 10+ technical patents and 50+ professional solutions.
As a council member of the Guangdong Clean Industry Association, we hold the Guangdong Province "Contract-Abiding and Trustworthy" enterprise certification, along with qualifications for building decoration engineering, building M&E installation engineering, and medical device business licensing.
What we offer phytic acid sodium manufacturers is not just a cleanroom, but a complete verifiable, traceable, and export-compliant full-process environmental assurance system.
Is Your Phytic Acid Sodium Workshop Facing the Same Pressures?
If you're experiencing microbial contamination returns, product moisture absorption and clumping, heavy metal indicators near limits, or inability to provide export compliance documentation...
Contact Keyao to receive a free "Phytic Acid Sodium Cleanroom Construction Plan". Our engineers will provide targeted renovation recommendations based on your existing workshop conditions and product standards.
13 years of focus, 100+ verified clients — Guangdong Keyao, worthy of your trust.

☎️ Phone: 13929950401
WeChat: Follow the "广东科耀" official account or scan the QR code below to add WeChat

Website: www.gdforyou.com
📊 Project Results Showcase

About Guangdong Keyao
Guangdong Keyao Purification M&E Engineering Co., Ltd. — 13 years of cleanroom engineering expertise, serving 100+ clients with 10+ technical patents.
Three professional domains: Industrial Cleanrooms (plant extracts, biopharmaceuticals, health supplements, food & cosmetics), Medical Cleanrooms (operating theaters, ICUs, laboratory departments), and Laboratory Cleanrooms (PCR, microbiology labs, temperature & humidity controlled labs).
Guangdong Clean Industry Association Council Member | Guangdong Province "Contract-Abiding and Trustworthy" Enterprise | Building Decoration Engineering Professional Contracting Qualification
Protecting phytic acid sodium production from microbial, moisture, and heavy metal risks — this is our commitment to every plant extract cleanroom we build.

